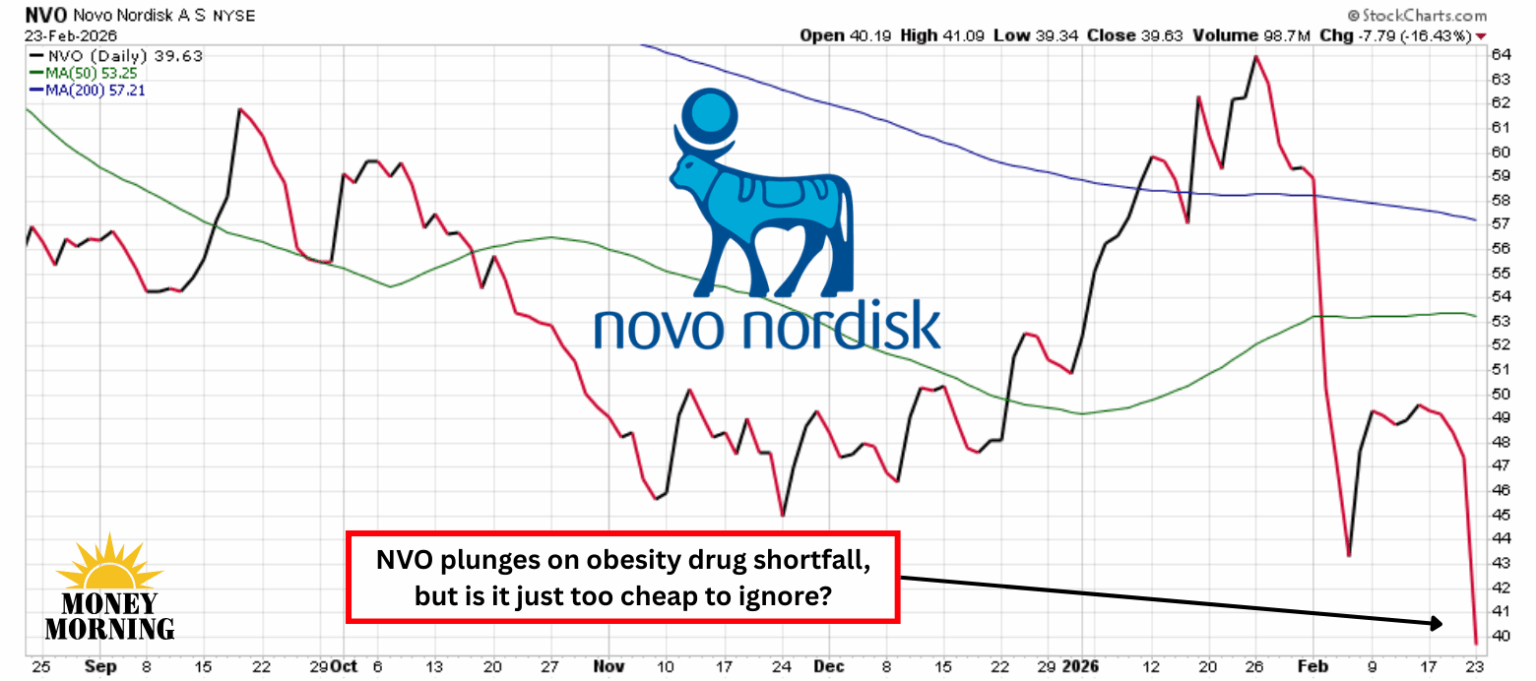
Novo Nordisk (NVO) once dominated the weight-loss revolution with Wegovy and Ozempic, but Eli Lilly (LLY) has seized the lead with Zepbound and its pipeline momentum. The Danish giant’s latest bid to regain momentum – CagriSema – came up short in a high-stakes head-to-head trial, triggering a brutal 16% stock plunge to levels not seen since 2021.
Investors are rattled, fearing Novo is ceding the $100-billion-plus obesity market. Yet for patient, long-term investors, this sharp selloff may represent a classic “buy the fear” moment in a company with deep scientific roots, proven commercial muscle, and multiple shots still on goal.
Is It As Bad As All That?
CagriSema – Novo’s dual GLP-1/amylin agonist – delivered a solid 23% average weight loss over 84 weeks in the open-label REDEFINE-4 trial versus tirzepatide’s 25.5%. On the stricter treatment-policy definition, the gap widened to 20.2% versus 23.6%, narrowly missing the statistical bar for non-inferiority. The market’s reaction was severe as investors interpreted the miss as proof Novo cannot catch Lilly’s dual-agonist juggernaut.
Wall Street also appears ready to wave the white flag. Deutsche Bank’s Emmanuel Papadakis downgraded the stock from Buy to Hold and slashed his price target by 31%, the second time in two weeks he has reduced his target.
 But are the results really that catastrophic? Not when viewed in context. CagriSema fell just short in a head-to-head setting against the current gold standard – yet it pursued a genuinely novel mechanism by pairing semaglutide with cagrilintide, an amylin analog that mimics a different satiety hormone.
But are the results really that catastrophic? Not when viewed in context. CagriSema fell just short in a head-to-head setting against the current gold standard – yet it pursued a genuinely novel mechanism by pairing semaglutide with cagrilintide, an amylin analog that mimics a different satiety hormone.
Amylin works on brain pathways distinct from GLP-1/GIP, potentially offering better appetite control, muscle preservation, or efficacy in patients who plateau or respond poorly to tirzepatide alone. Early signals from prior REDEFINE studies already hint at differentiated benefits for certain subgroups.
Importantly, Novo never positioned CagriSema as a direct “me-too” replacement. Management has repeatedly framed it as an option for patients who do not achieve sufficient results on Zepbound or Wegovy. That niche strategy remains intact. The company is pressing ahead with optimized dosing in the REDEFINE-11 trial and other studies to refine the regimen and potentially close any efficacy gap.
A Glimmer of Hope
Yet, one day after the CagriSema disappointment, Novo delivered fresh positive news on its next-generation candidate. UBT251, the triple agonist (GLP-1/GIP/glucagon) licensed from United Biotechnology and designed to challenge Lilly’s retatrutide, posted impressive Phase 2 data from China: up to 19.7% mean weight loss at 24 weeks versus just 2% for placebo.
That edges out Lilly’s retatrutide’s 17.5% at the same time point in its own Phase 2 trial – though retatrutide later reached 24.2% at 48 weeks. UBT251’s global Phase 1b/2a data are due in 2027, with Phase 3 to follow. It’s early, but the trajectory is encouraging and underscores Novo’s commitment to staying ahead in the evolving “triple G” race.
Bottom Line
The CagriSema miss was undeniably disappointing and justified some near-term pain. Yet it is far from the devastating blow the market reaction implies. CagriSema still produced clinically meaningful weight loss with a novel therapy that could still shine as development continues.
Meanwhile, UBT251’s strong early readout keeps Novo firmly in the hunt. After the selloff, Novo Nordisk trades at an historically low PE of 11 and 15x free cash flow. For a company with blockbuster franchises, robust cash flow, and a deep pipeline, this stumble may ultimately prove to be an attractive entry point for patient investors willing to look past the headlines.
— Rich Duprey
$3 billion+ in operating income. Market cap under $8 billion. 15% revenue growth. 20% dividend growth. No other American stock but ONE can meet these criteria... here's why Donald Trump publicly backed it on Truth Social. See His Breakdown of the Seven Stocks You Should Own Here.
Source: Money Morning
